24小时服务热线
13007546599
服务热线:13007546599
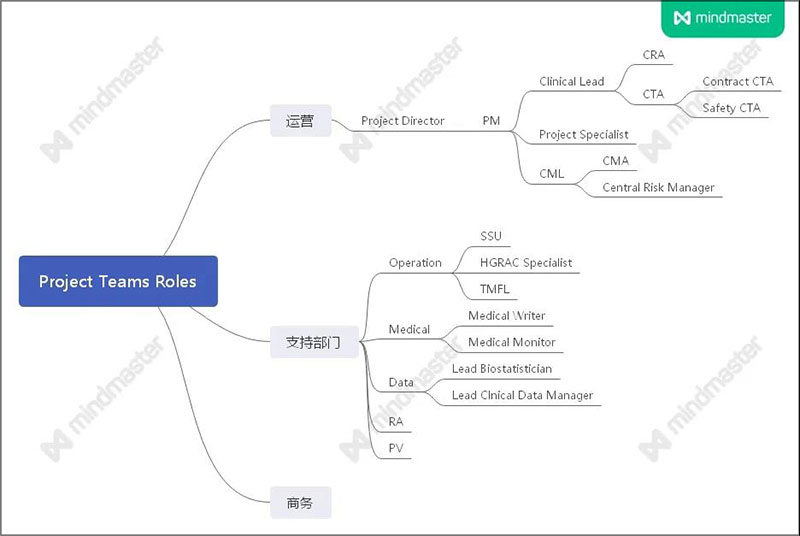
部分名词说明
Project Director(PD)
Opreates as a point of escalation for both the PM and sponsor
Project Manager(PM)
Responsible for deliverling the project in accordance with the contract and the protocol ensuring compliance with regulations
Clinical Lead (CL)
Liaise with PM and CRAs,manages clinical aspects of project, primary contact for CRAs and primary in house contact for sites. Also liaises with the Project Specialist(PS)
Project Specialist(PS):PM's Left Hand
1.Responsible for utilizing CTMS system to track and analyze trial information(e.g. site selection,enrollment,ICF dates etc.). Will provide updates and reports to Project Leadship
2.Participate in Kick-Off Meetings, takes meeting highlights and follows up with team on action items
3.Senior PS can take the role as Clinical Lead
Trial Master File Lead (TMFL)
Provide oversight of ball electronic and/or paper TMF management activities for assigned projects and/or progarams from award to final transfer
Lead Biostatistician
writes and reviews statistical Analysis Plans (SAP) based on protocol, including development of well-presented mock-up displays for Tables,Listing and Figures (TLF)
Project Financial Analyst
Records revenue each month for Unit, Fee for service and fixed price contracts.
Medical Writer
Complies, writes and edits medical writing deliverables under the guidance for research scientist including: Protocols , informed Consent Form and more
Lead Clinical Data Manager(LCDM)
Receives verified data from CRA, looks at trends in the data, submit queries to sites
Study start-up
Performs assigned activities within the country;start-up of investigator sites in all phases of clinical trial
Central Monitor(CMA)
1.Foucus on data quality and reporting
2.Collaborate with CRA on guidance outlined in the clinical monitoring plan
Central risk manager
1.Facilities the risk assessment process
2.Coodinates cross-functional data review strategy
当然,还有部分职位在上图中没有体现出来,欢迎大家留言区补充啦!!!
站点声明:
本网站所提供的信息仅供参考之用,并不代表本网赞同其观点,也不代表本网对其真实性负责。图片版权归原作者所有,如有侵权请联系我们,我们立刻删除。如有关于作品内容、版权或其它问题请于作品发表后的30日内与本站联系,本网将迅速给您回应并做相关处理。
河南岁福医疗科技有限公司专注于医疗器械、诊断试剂产品政策与法规规事务服务,提供产品注册申报代理、临床合同(CRO)研究、GMP质量辅导等方面的技术外包和生产许可证、经营许可证及临床机构备案办理服务。

相关文章